BeH 2, on the other hand, may be created by the reaction of BeCl 2 with LiAlH 4 2BeCl 2 + LiAlH 4 → 2BeH 2 + LiCl + AlCl 3.Except for beryllium, other elements interact with hydrogen to generate their hydrides, MH 2.BeO + C + Cl 2 -600 to 800 K→ BeCl 2 + CO Thermal decomposition of (NH 4 ) 2 BeF 4 is the best method for producing BeF 2, and BeCl 2 is easily produced from the oxide.M + X 2 → MX 2, where X represents the halogens, including fluorine, bromine, chlorine, and iodine. 
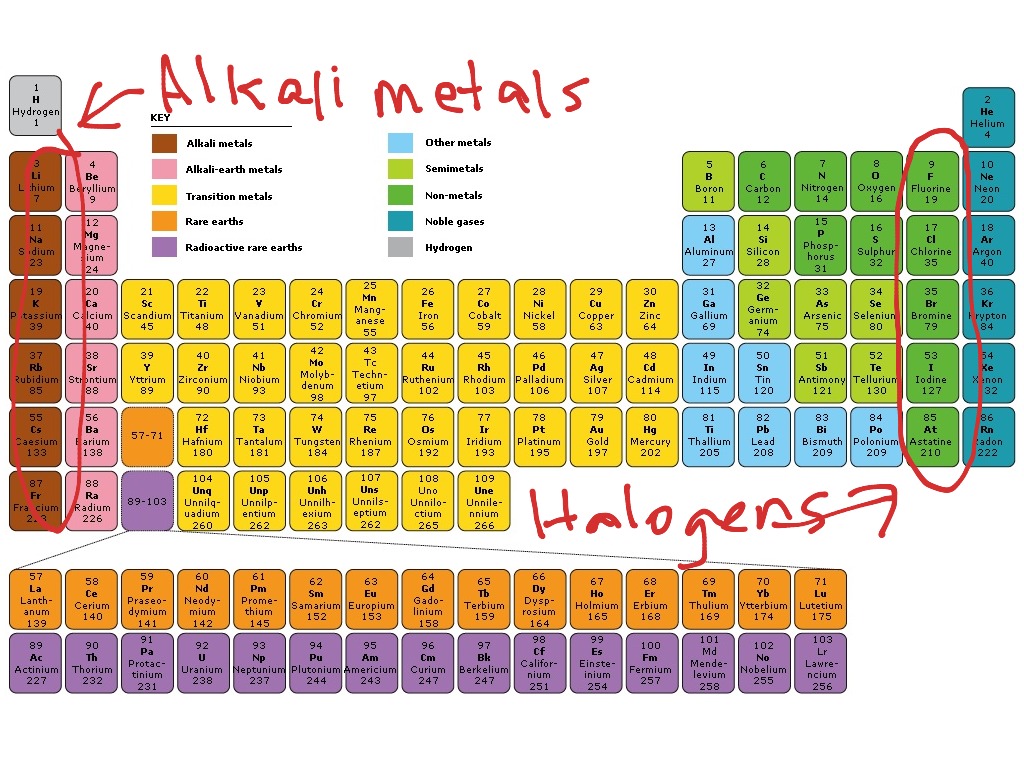
At high temperatures, most alkaline earth metals interact with halogen to generate their halides.These also react with intense water, even in freezing temperatures, to produce hydroxides. Air quickly affects calcium, strontium and barium to generate oxide and nitride.Magnesium is highly electropositive and burns brightly in the air to produce MgO and Mg 3 N 2.Powdered beryllium, on the other hand, burns excellently in air when ignited, producing BeO and Be 3 N 2.Because of the creation of an oxide coating on their surfaces, beryllium and magnesium are unresponsive to oxygen and water, so they do not react at all.Let us look at the different aspects of reactivity of alkaline earth metals: Reactivity with water and air As we move down the group, the reactivity of such elements rises. Alkaline earth metals have a lower reactivity than alkali metals.These elements’ compounds, just as alkali metals, are mostly ionic.Their basic electronic configuration is denoted as ns2. These elements contain two electrons in the valence shell s-orbital.The first element, beryllium, contrasts from the others in its diagonal interaction with aluminium.Except for beryllium, these are known as alkaline earth metals. In the periodic table, they come after alkali metals.Beryllium, magnesium, calcium, strontium, barium and radium are elements in Group 2.Beryllium and magnesium oxides and other compounds are more covalent than those generated by the heavier and larger components (Ca, Sr, Ba). This happens because of the greater nuclear charge and smaller size. The alkaline earth metals create mainly ionic compounds but are less ionic than the equivalent alkali metal compounds. Group 2 elements have the dipositive oxidation state ( M 2+) as their primary valence. These elements are abundant in the earth and thus acquire this name. The elements occurring in group 2 of the periodic table are alkaline earth metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
